May 15, 2026
Can sustainable polymers truly combine high performance with circularity? 🌿
At Avantium, we are developing PEF (polyethylene furanoate), known under the brand name releaf® by avantium, a next-generation polyester produced from the bio-based building block 2,5-furandicarboxylic acid (FDCA).
🌟 Compared to conventional fossil-based PET, PEF offers significantly improved barrier properties against oxygen and CO₂, together with improved moisture barrier performance. Fully recyclable, it enables applications across rigid packaging such as bottles, films, and textiles.
❓ Curious to learn more about this new high-performance polyester?
Join my talk at the Bordeaux Polymer Conference (BPC 2026), taking place from 1–4 June in Bordeaux, France, bringing together more than 500 delegates from academia and industry.
🎤 Talk title:
👉 Effect of diethylene glycol (DEG) on the crystallization kinetics of poly(ethylene furanoate) (PEF)
📅 03 June | 16:05
🟡 Looking forward to engaging in scientific discussions and exploring collaboration opportunities with researchers, academia, and industry experts working on the future of sustainable polymer materials.
🔗 BPC2026 website: Bordeaux Polymer Conference 2026

May 08, 2026
Have you heard about #PEF 🌿?
Is it really possible to have a 100% plant-based polyester with superior barrier performance?
🟢 Yes — PEF (polyethylene furanoate), with the brand name releaf® by avantium, produced from the bio-based building block 2,5-furandicarboxylic acid (FDCA), is exactly that. Derived from renewable plant-based sources, it offers a sustainable alternative to its fossil-based counterpart, PET, while delivering significantly improved barrier properties against oxygen, CO₂, and moisture. It is recyclable and designed to be compatible with existing polyester (PET) recycling streams and is driving innovation in high-performance, sustainable packaging. These properties enable applications ranging from rigid packaging such as bottles, to films and textiles.
I’m pleased to share that I’ve been invited as a speaker at the Milan Polymer Days International Congress (MIPOL 2026), taking place from 1–3 July in Milan, Italy.
🎤 Lecture title: Engineering crystallization kinetics in poly(ethylene furanoate) (PEF) through trace-level modulation
📅 July 02 | 10:05 AM
✨ I’ll be sharing what we’re building at Avantium—join the conference to connect and explore collaboration opportunities in sustainable materials innovation.
Looking forward to engaging with fellow innovators and experts in Milan.
🔗 MIPOL2026 website: Milan Polymer Days

May 05-06, 2026
Today, Plastics Recycling Show Europe 2026 (#PRSE2026) in Amsterdam, The Netherlands brought together a strong mix of ideas, perspectives, and 500+ exhibiting companies, creating momentum around the circular use of plastics, including polyesters.
Sharing a few photos from the event, including highlights from several insightful talks.
Clear progress in recycling technologies and polyester innovation within broader polymer systems, with scalability, feedstock quality, and system integration remaining key factors for broader implementation across the value chain.
October 07, 2025
🌱 Continuous learning: the passport that fuels growth across borders and teams.
Over the past 7–8 years, my journey through academia and now the R&D industry across Europe has taught me that innovation isn’t only about brilliant ideas—it’s about people. It thrives when diverse teams unite around a common purpose.
To strengthen this mindset, I recently completed several PMI-certified LinkedIn Learning courses:
✅ Building High-Performance Teams
✅ Managing Teams
✅ Stakeholder Management for Leaders and Managers
I strive to use these insights to bridge the gap between science and real-world impact, empower and unite diverse teams, and align stakeholders.
💡Continuous learning is an investment that always pays off. It shapes not only personal growth but also the success of teams and organizations.
December 16, 2024
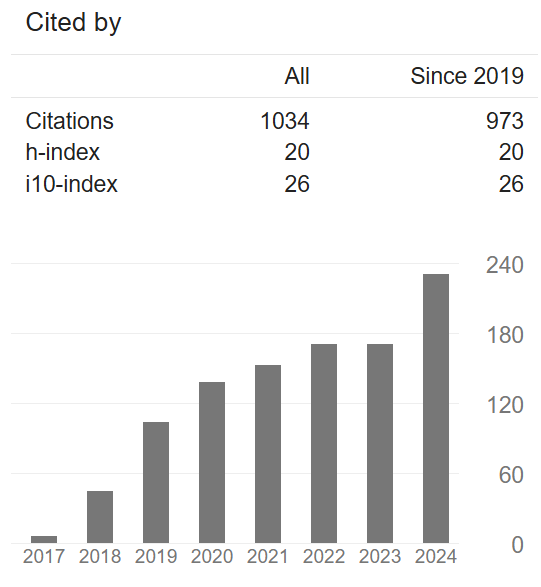
Thrilled to share that my published research work on the development of fully biobased polyesters from renewable resources, with a specific focus on those derived from 🟢sugars-based 2,5-furandicarboxylic acid (FDCA), have surpassed 1000 citations on Google Scholar with a h-index of 20!
👉This milestone would not have been possible without the opportunity given to me by Prof. Dimitrios Bikiaris and Prof. George Papageorgiou during my PhD study and Postdoc at Aristotle University of Thessaloniki in Greece to work on such a hot research topic: 🍀Design, synthesis and study of renewable furan-based polyesters for use as sustainable packaging materials, with great interest and in-depth investigation 👨🔬 into the 💯 biobased Polyethylene furanoate polyester (PEF)🌿, FDCA-based polyester blends, as well as new furanoate copolyesters derived from renewable monomers (isosorbide, vanillic acid, FDCA, among others…).
✔️I am also thankful to Prof. Minna Hakkarainen for hosting me in her labs at KTH Royal Institute of Technology in Stockholm_Sweden to work on very interesting polyester-related research topics, in particular:
👉 Open-loop chemical recycling of post-consumer polyester _like PET_ into new closed-loop chemically recyclable materials, thus supporting a circular plastics economy♻️..
✅In the same context, Polyethylene furanoate polyester (PEF)🌿 which has been launched by Avantium under the brand name Releaf is the next-generation renewable furan-based material with exceptional properties: a recyclable, 100% plant-based polyester ideal for the manufacture of bottles, films and fibres.
Avantium is set to start the commercial production of releaf® by avantium in 2025🎉!
🌿Let’s move towards a fossil-free chemical industry, especially after the official opening of the world’s first commercial scale FDCA Flagship plant of Avantium in Delfzijl, the Netherlands.
My google scholar account: LINK
October 24, 2024
This week, it was a very special and memorable moment to celebrate 🎉 the official opening of Avantium’s world’s first commercial scale FDCA Flagship plant in Delfzijl, the Netherlands 👉 A remarkable step forward in the world of sustainable materials 🍀!
✅2,5-furandicarboxylic acid (FDCA) is the building block used to make polyethylene furanoate polyester (PEF) which has been launched under the brand name Releaf: a recyclable, 100% plant-based polyester ideal for the manufacture of bottles, films and fibres.
Avantium is set to start the commercial production of Releaf in 2025!
🌿Let’s move towards a fossil-free chemical industry!
April 3-5, 2024
Together with my colleague Roy from Avantium we participated in the FUR4Sustain COST Action conference held in Nice – France from April 3 to 5.
✅The three-day meeting was full of interesting talks and discussions on bio-based polymers derived from 2,5-furandicarboxylic acid (FDCA), including next-generation recyclable polyester PEF – the renewable-based solution to its fossil-based counterpart polyethylene terephthalate (PET).
A special thanks to Dr. Nathanael Guigo for organizing and hosting such a great event at the Université Côte d’Azur.
It has been a pleasure to be part of the FUR4Sustain network over the past four years!
February 01, 2024
I am very delighted to be one of 24 young scientists, assembled from across 4 continents, to join the ACS Journal Biomacromolecules as members of the new Early Career Board. 🎊📣🔔
✅ This year, the journal Biomacromolecules (Impact factor: 6.2) published by the American Chemical Society celebrates its 25th anniversary. It is one of the most recognized forums for the dissemination of cutting-edge research in polymer science and biology, led by Professor Sebastien Lecommandoux (Editor-in-Chief).
🔗 For more details, please visit the links below:
Journal website: Here
Editorial link: Here / PDF file: Here

December 01, 2023
I’m happy to share that I’m starting a new position as R&D Project Leader / Polymer Scientist at Avantium in Amsterdam, Netherlands.
Activities: Development of biobased polyesters from 2,5-furandicarboxylic acid (FDCA), including PEF.
November 02, 2023
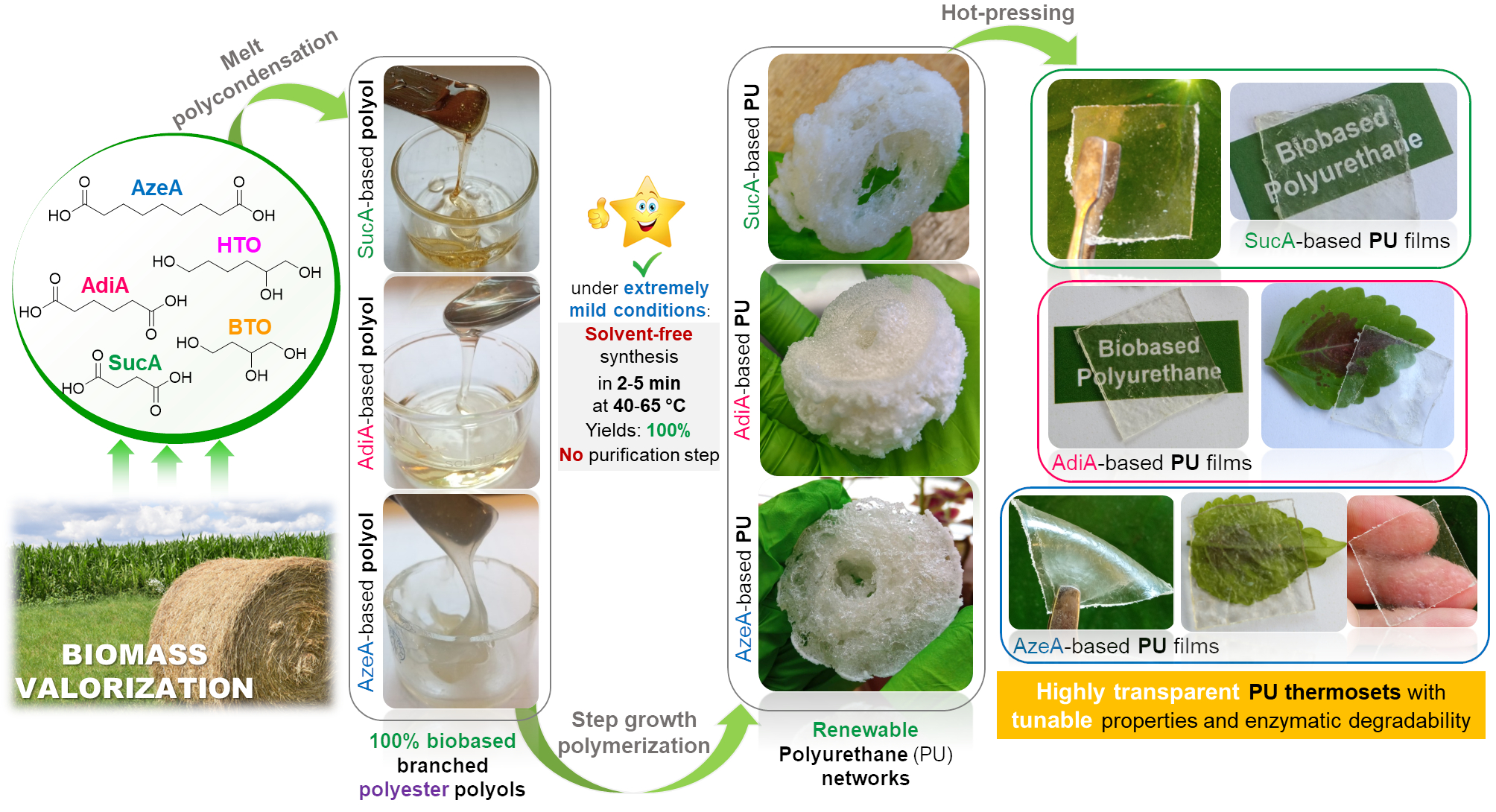
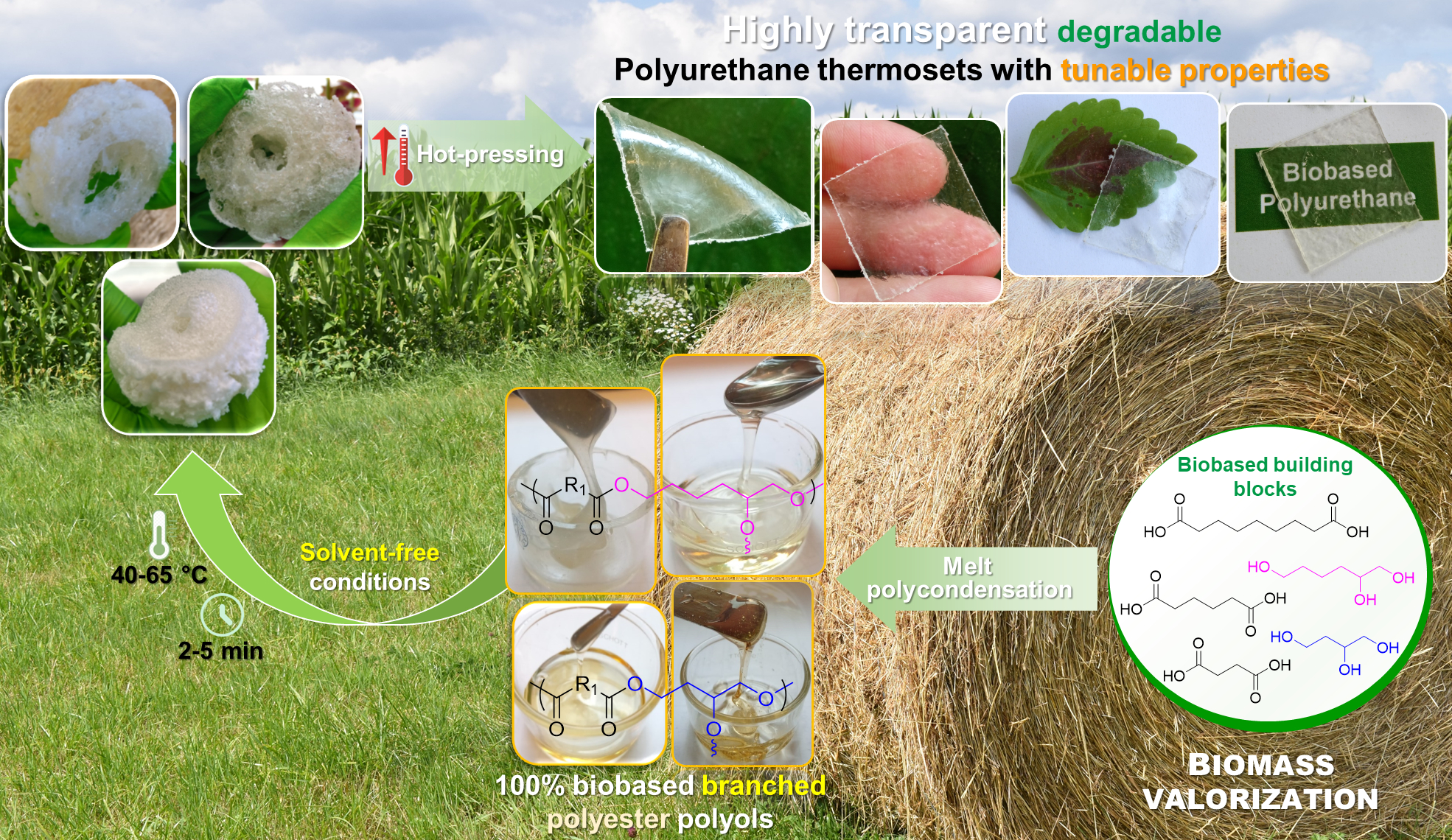
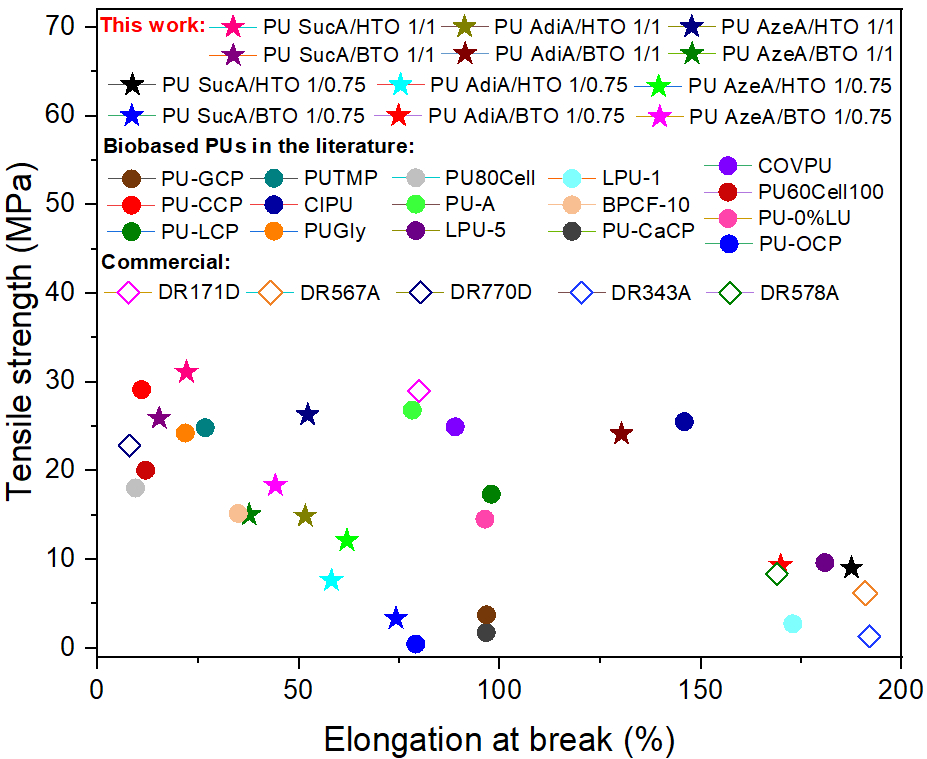
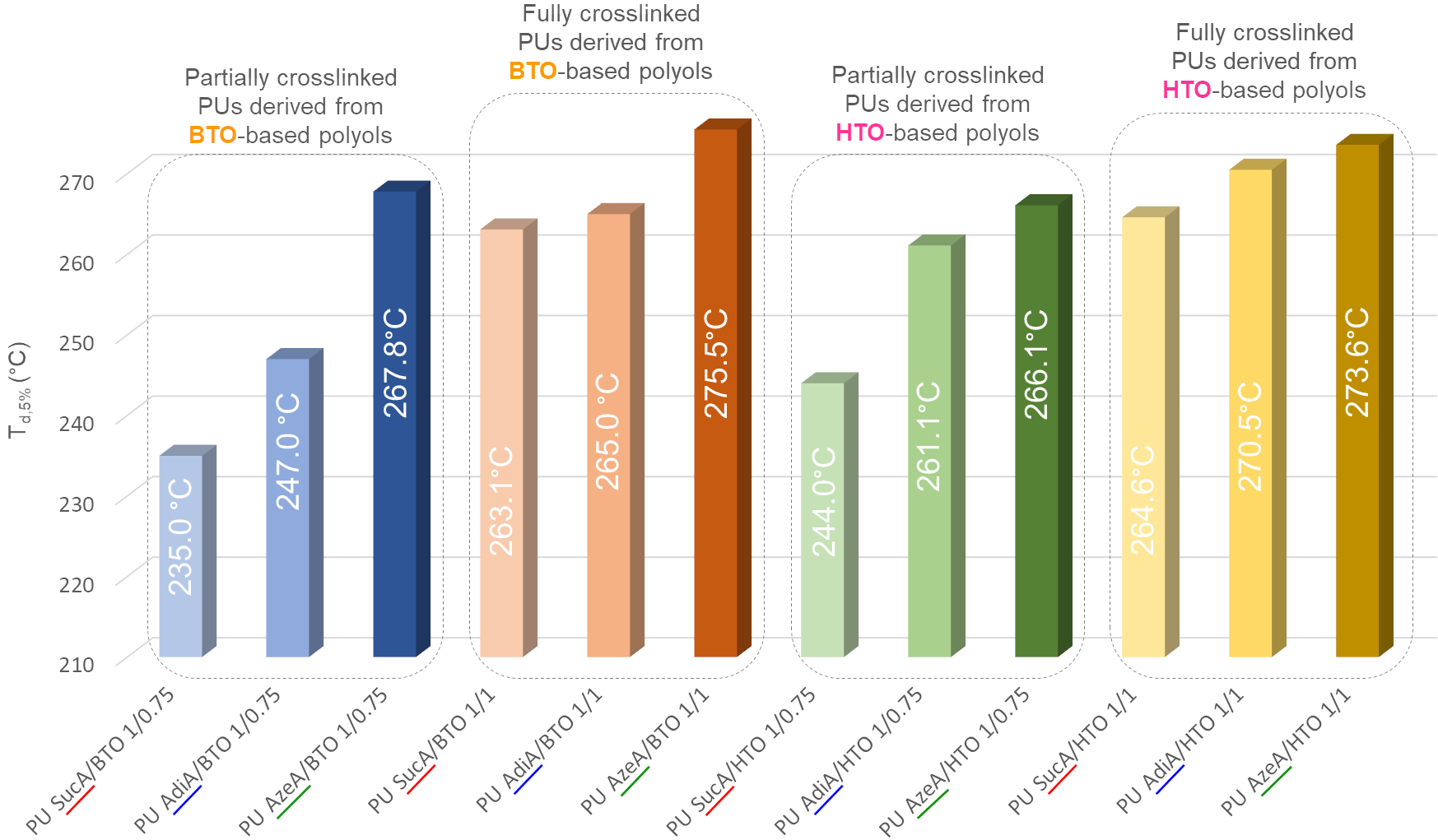
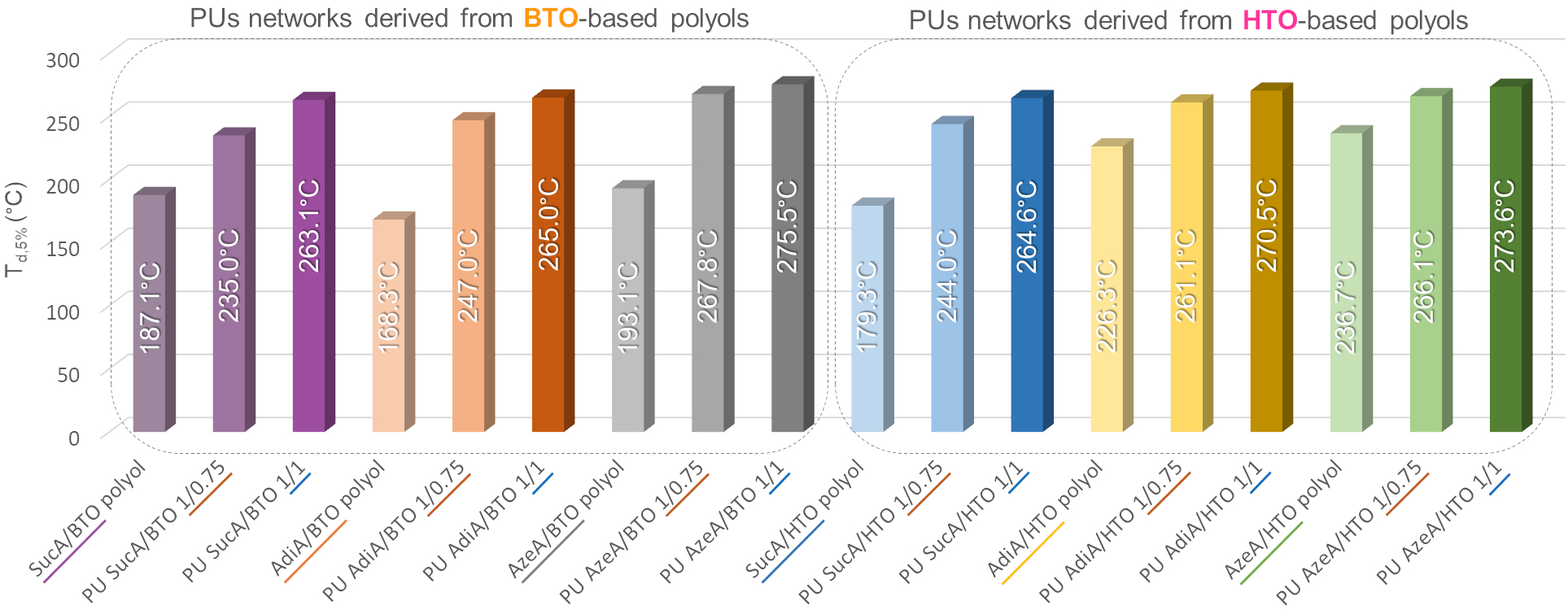
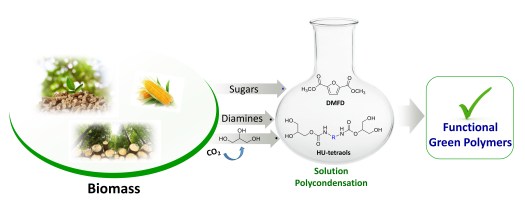
🤔Are you interested in exploring a novel formulation of fully bio-based branched polyester polyols from hemicellulosic sugars as well as their use in the synthesis, using a highly sought synthetic approach for industrial scalability, of highly transparent polyurethane (PU) thermosets?
✅The resulting innovative PU materials, with readily tailorable thermal and mechanical properties and enzymatic degradability, were developed by solvent-free step growth polymerization under extremely mild reaction conditions (125-335 s, 40-65°C and yields of 100%!!) and showed a very promising applications spectrum!!
👉You can find the detailed answer here in the full OPEN ACCESS 🔓article recently published in Green Chemistry: LINK
⚠️ In the videos below👇, fully crosslinked transparent PU films derived from biobased monomes are shown as representative examples:
October 27, 2023
I am thrilled to share with you my latest paper published in the prestigious Royal Society of Chemistry journal – Green Chemistry (IF: 9.8).
The title of the OPEN ACCESS article : “Highly transparent polyurethane thermosets with tunable properties and enzymatic degradability derived from polyols originating from hemicellulosic sugars“
In this work, a novel portfolio of highly transparent aliphatic polyurethane thermosets, synthesized under very mild conditions from 100% renewable branched polyesters, exhibiting “on demand” tunable mechanical and thermal properties and enzymatic degradability.
For more details, visit the publication link : https://doi.org/10.1039/D3GC03302C
Thanks Yosra Chebbi, Prof. Alessandra Lorenzetti and Prof. Minna Hakkarainen for your priceless contribution to this excellent study!
KTH Royal Institute of Technology
April 28, 2023
My latest paper “Open-loop recycling of post-consumer PET to closed-loop chemically recyclable high-performance polyimines” published in Resources, Conservation & Recycling journal (IF: 13.716) was selected by the editors as the Cover article of the volume 193, June 2023!!
Publication link: https://lnkd.in/enjinVtz

March 27, 2023
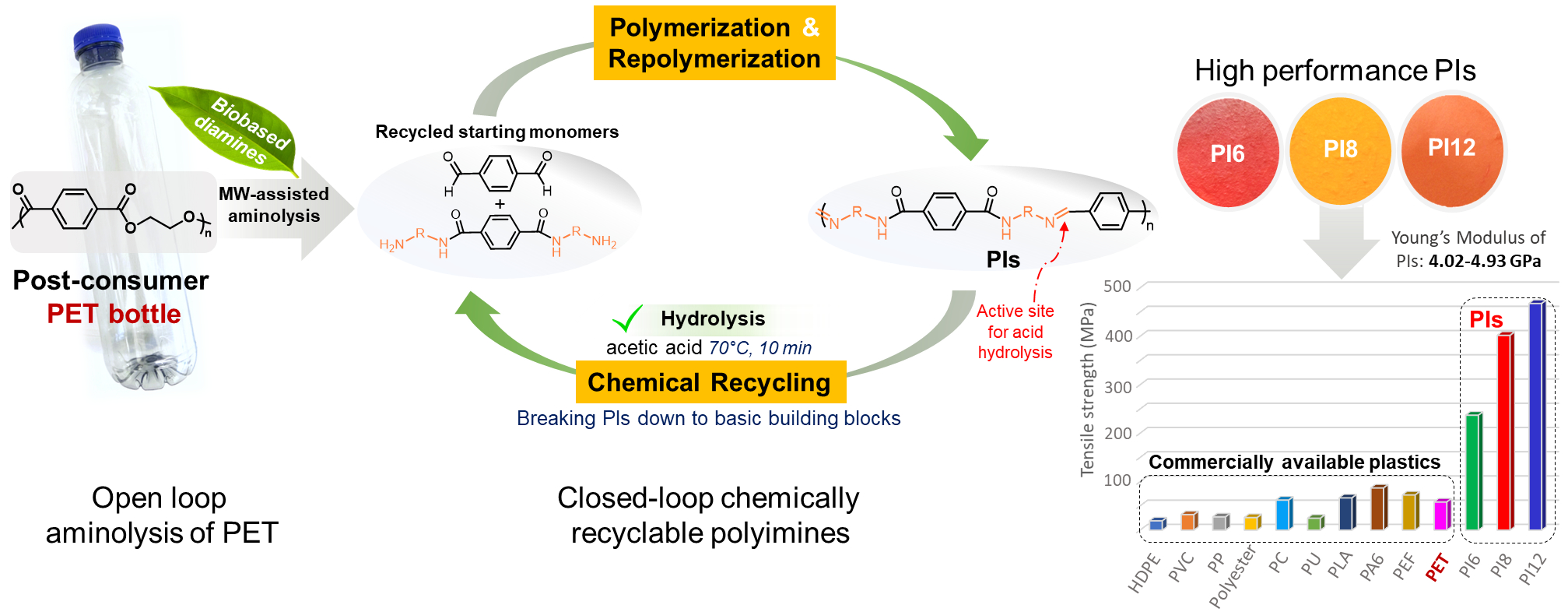
Can chemical recycling play an important role as a future end-of-life option for turning post-consumer PET bottles into very promising closed-loop recyclable polymers, thereby supporting the circular plastics economy?
The detailed answer is here in our latest contribution, recently published in the prestigious Elsevier Resources, Conservation and Recycling journal (IF: 13.716, rank: 3/110 in “Waste Management and Disposal” category).
The title of the OPEN ACCESS article : “Open-loop recycling of post-consumer PET to closed-loop chemically recyclable high-performance polyimines“
For more details, visit the publication link below: https://lnkd.in/enjinVtz
The graphical abstract is found here: https://lnkd.in/eHZ9MDWN
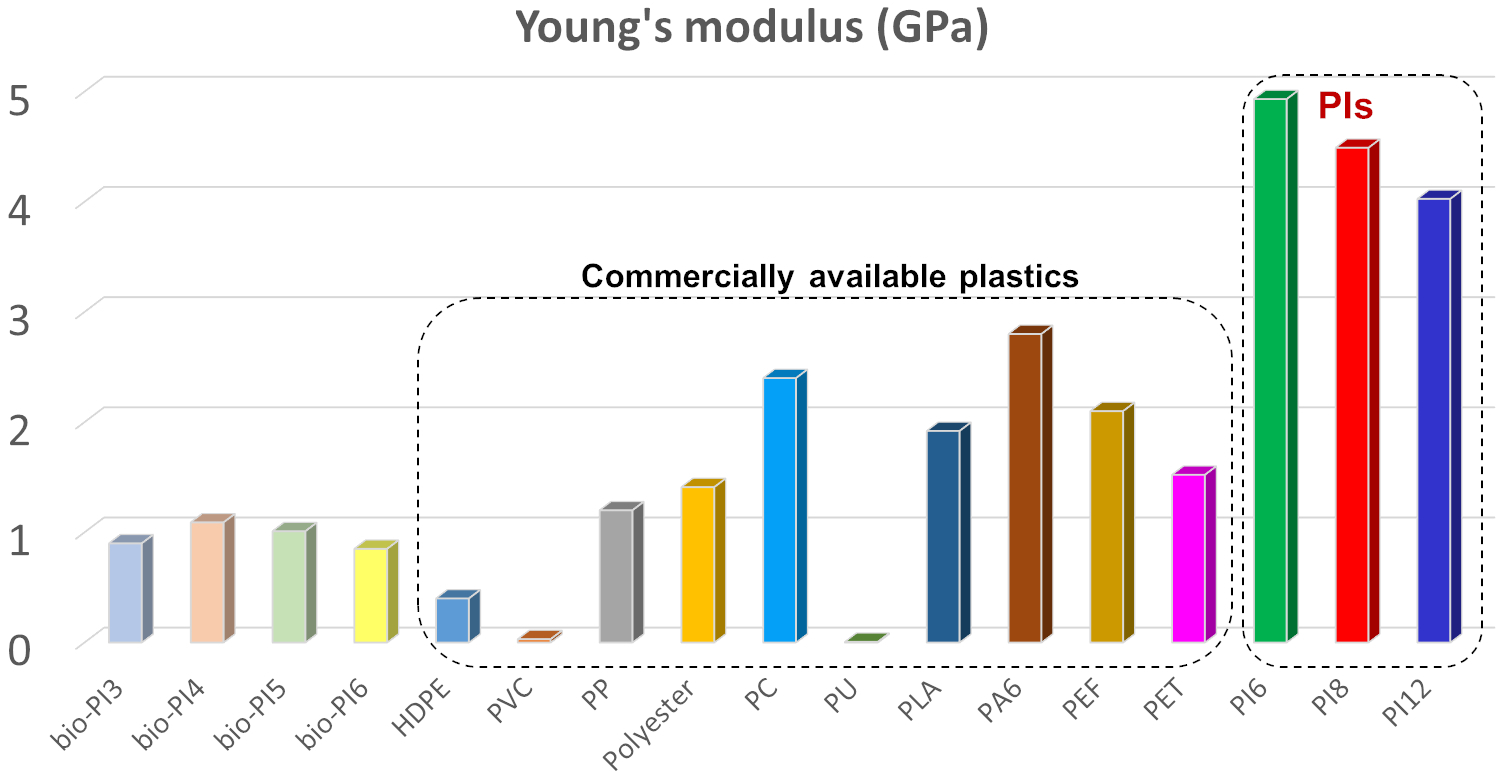
In this work, intriguing high-performance polyimines (PIs) were designed from diamines recovered by open-loop recycling of postconsumer PET bottles by microwave-assisted aminolysis. These new PIs had excellent thermal properties, super-high toughness and strength, very impressive strength, with Young’s modulus of 4.02-4.93 GPa and tensile strength of 237-467 MPa, significantly outperforming most of available commodity plastics in the market!! They robust PIs demonstrated recyclability “on demand” to original building blocks via mild and ultrafast acetic acid catalysed hydrolysis. The recovered building blocks could be directly repolymerized back to PIs with similar properties to the original PIs, thus avoiding any additional separation process of the recycled products!! Taking advantage of their features, the newly developed PIs thermoplastics show great potential to serve as next-generation circular materials, designed from recycled plastic waste, for a wide range of practical applications.
Excited to share our findings with the academic and industrial community!!
Thanks Eva Bäckström and Prof. Minna Hakkarainen for collaborating and for your priceless contribution to this so pleasant recycling work!
KTH Royal Institute of Technology
Keywords: PET, packaging, Chemical recycling, aminolysis, Schiff-base, Dynamic covalent chemistry, Polyimine, Circular plastics economy, plastics, waste management, plastics recycling
September 13, 2022
Work in progress:
Development of a new biobased portfolio of biodegradable polyurethane thermosets derived from renewable feedstocks with “on demand” tunable thermal properties and mechanical performance (7.3< tensile strength (MPa) <33.4 and 15.6< strain (%) <189), and optical transparency for very promising practical applications…
June 29, 2022
Research is currently underway in our labs to develop a new series of biobased polyester-urethane (PU) thermosets with on-demand tunable properties!! The solvent-free synthesis process of these biodegradable cross-linked PU networks involves the use of new sustainable branched polyols featuring unique macromolecular architectures made 100% from biomass, particularly hemicellulosic sugars. The polymerization was successfully carried out under mild conditions (< 50 °C) and took only 5 min!! Let’s move on together to greener materials in the future!!
June 11, 2022
Our contribution to the development of the renewable thermoplastic polyester material, poly(lactic acid) PLLA, was recently published in International Journal of Biological Macromolecules (IF: 8.025 / Q1), in collaboration with Prof. Alejandro J. Müller from the University of the Basque Country UPV/EHU, Spain.
The title of the open access article: “Effect of the structural features of biobased linear polyester plasticizers on the crystallization of polylactides”
In this work, three new 100% biobased polyester plasticizers derived from adipic acid (AdA) were successfully employed, for the first time, to accelerate the crystallization rate and enhance the mechanical properties of polylactide (PLLA); an important step to improve its performance and expand its range of applications.
For more details, visit the link below: https://doi.org/10.1016/j.ijbiomac.2022.06.056
The addition of AdA-based plasticizers to PLLA:
> significantly increases the elongation at break up to 8 times compared to neat PLLA.
> increases the nucleation rate from the glassy state by around 50-110 %.
> causes a remarkable increase in the overall crystallization rate from the glassy state which was 2-3 times faster for the plasticized PLLAs than neat PLLA.
> decreases Tg by up to 11 ºC
Such findings make these aliphatic polyester plasticizers derived from fully renewable resources very promising for improving the properties and applications of PLA.
June 1-3, 2022
This week I presented my work entitled “Designed from Recycled PET packaging waste” at Nordic Polymer Days 2022 Conference in Gothenburg, Sweden. It has been a great opportunity to discuss with many inspiring scientists.
In this work, an efficient and fast end-of-life management of non-degradable poly(ethylene terephthalate) thermoplastic waste by chemical recycling was designed. The approach involves transforming PET waste, exemplified by a post-consumer PET bottle, by microwave-assisted solvent-free aminolysis into new value-added terephthalate diamines featuring high melting points. These monomers were then used to synthesize new aromatic-aliphatic high Tg polyimines.
Transforming post-use terephthalate packaging into new high-performance monomers and polymers with outstanding performance, rather than sending them to landfill or incineration, is a much-needed step to prevent generation of packaging waste and to reduce the fatal effects of plastic pollution on our planet.
December 20, 2021
Our latest contribution to the development of high-performance Furanoate bioplastics was just published in Polymer Degradation and Stability (IF: 5.204 / best quartile journal Q1).
Article title: “Tuning thermal properties and biodegradability of poly(isosorbide azelate) by compositional control through copolymerization with 2,5-furandicarboxylic acid“
In this work, a novel fully biobased FDCA-based copolyesters series, distinguished by improved thermal performance (tunable Tg over a high and broad temperature window oscillating from 9 to 91°C depending on FDCA content), sustainability and enhanced susceptibility to enzymatic degradation, was synthesized by melt polycondensation..
For more details, visit the link below: LINK
November 05, 2021
It is a great privilege for me to serve as Guest Editor of a new Special Issue in Polymers MDPI (Q1, IF: 4.967) themed “Development of High-Performance Biobased Polyesters” with Professor Dimitrios Bikiaris from Aristotle University of Thessaloniki, Greece (LINK) and Professor Minna Hakkarainen from KTH Royal Institute of Technology, Stockholm, Sweden (LINK)
For further reading, please follow the link of the SI: https://lnkd.in/eywmJXP
Deadline for manuscript submissions: 15 November 2022.
We welcome in advance your excellent contributions to this Special Issue by submitting original research articles, review articles, or short communications aligned with this topic.
November 01, 2021
It is time to move on and start a new chapter in my career as Researcher at one of Europe’s leading technical and engineering universities: KTH Royal Institute of Technology , Stockholm, Sweden

To be successful, the first thing to do is fall in love with your work !!







28 September 2021
I am delighted to attend the 5th European Conference on Green and Sustainable Chemistry – 5th EuGSC (26-29 September 2021), where I presented my latest work entitled: “Tuning thermal properties and biodegradability of Isosorbide-based polyester by compositional control through copolymerization with 2,5-furandicarboxylic acid“
These innovative copolyesters, distinguished by improved thermal performance, noticeable susceptibility to enzymatic degradation, and sustainability are highly desired. These unique characteristics endow this novel class of furan-based copolyesters a great potential for a broad range of practical applications requiring high Tg values and make them a concrete contribution towards greener materials…




Coming soon!! 14th & 15th September 2021: FUR4Sustain summer meeting Agenda
Get ready for two days of interesting talks, knowledge and networking!
It is with great pleasure that we all will meet (in-person or on-line) in the next 14th and 15th of September to discuss and advance the future of Furan-based materials, in particular, and sustainable polymers, in general.
Please find below the agenda for these two exciting days!
22 July 2021
COST network wants to introduce innovative and sustainable furan-based plastics on the market
Did you know that many alternatives exist to move-away from unrenewable fossil-resources-based plastic as we use it today?
Different biomass-derived polymers are possible and could be introduced on the market, but challenges remain as research and initiatives are scattered.
COST Action FUR4sustain brings together experts from multidisciplinary backgrounds and looks at innovative solution such as furans for green alternatives.
For more details, click here
#sustainability #FUR4sustain #2,5-FDCA #furans #polymer #greenplastic #researchnetwork #environment
29 March 2021
We are pleased to share our latest publication on the development of renewable Furan-based polyester blends, published by BIKIARIS Group @ Polymers (Q1, IF=4.329).
Article title: “A New Era in Engineering Plastics: Compatibility and Perspectives of Sustainable Alipharomatic Poly(ethylene terephthalate) /Poly(ethylene 2,5-furandicarboxylate) Blends“
For more details, visit the link below:
https://lnkd.in/ebwA2uz
In this work, a series of Poly(ethylene terephthalate)/poly(ethylene 2,5-furandicarboxylate) polyester blends in the entire range of compositions have been successfully prepared. The structure, thermal transition temperatures and the thermodynamics of miscibility phenomena of the synthesized Furanoate blends have been studied in detail…
To sum up, such blends are expected to play a major role in the near future for the increase in sustainability and the transition to green economies.
02 March 2021
One of our outstanding contributions to the development of fully biobased furanoate polyesters has just been published yesterday by BIKIARIS Group in Green Chemistry (IF=10.182).
Title of our publication: “Investigation of the catalytic activity and reaction kinetic modeling of two antimony catalysts in the synthesis of poly(ethylene furanoate)“
For more details, visit the link below: (LINK)

21 January 2021 My latest paper has just been published today in Polymer Chemistry (IF=5.582).
Article title: “Synthesis and characterization of fully biobased polyesters with tunable branched architectures“
For more details, visit the link below: (LINK)

17-18 December 2020 I am delighted to attend the “FUR4Sustain XMAS meeting 2020“ (COST Action CA18220, LINK 1, LINK 2) #FUR4Sustain


06 October 2020 I am very delighted to join the “European network of FURan based chemicals and materials FOR a Sustainable development” (COST Action CA18220, LINK 1, LINK 2)

07 August 2020 In my latest study published in Polymer Degradation and Stability “IF=5.030” (click here), a truly efficient, practical, and more environmentally friendly solvent-free synthetic route was successfully applied herein to prepare three new fully biobased diol monomers derived from vanillic acid. To ascertain their stability toward the conditions of melt polycondensation, these diols were subjected to melt polycondensation with diacyl chlorides having different length alkane bridges and also with dimethyl furan-2,5-furan dicarboxylate.
The overall conclusion is that such work is a concrete contribution towards the valorization of vanillic acid. It can be used as a support and helpful reference to highlight the feasibility of a more environmentally benign solvent-free pathway for sustainable building blocks design.
This innovative synthesis route that could be readily applied at industrial scale makes the development of fully biomass-derived plastics easier and faster, which is the real willingness of both the scientific and industrial communities.
15-17 July 2020 It is delighted to give an oral presentation at virtual Milan Polymer Days conference (MIPOL2020) about our recent work entitled: “Solvent-free synthesis of new fully biobased diol monomers through industrially viable approach: Toward new insights into the valorization of vanillic acid-based polyesters” (Click here)

16 March 2020 For first time, the synthesis of a novel furanoate polyester-urethane with vitrimer-like behavior was reported in our lastest publication published in Biomacromolecules “IF=6.988” (Click here)
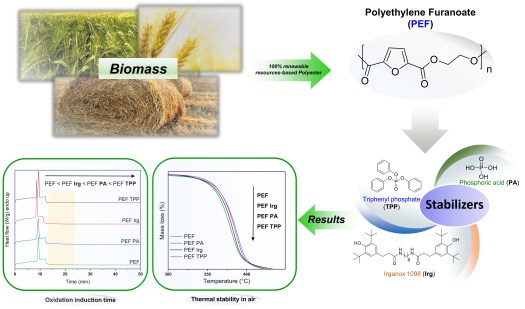
04 February 2020 In our recent study published in Thermochimica Acta (click here), the effect of three different stabilizers on the physicochemical properties of the poly(ethylene 2,5-furan dicarboxylate) (PEF) biobased polyester have been studied for the first time.
25 September 2019 My recent paper published in Polymer Degradation and Stability “IF=5.030” (click here), describing an effective solution for overcoming the low glass transition temperature (Tg) of poly(hexamethylene 2,5-furan dicarboxylate) (PHF), is proposed that uses isosorbide as a bicyclic rigid diol comonomer. Such kind of copolymers developed herein, which combines both the sustainability and improved thermal performance, is highly desired…
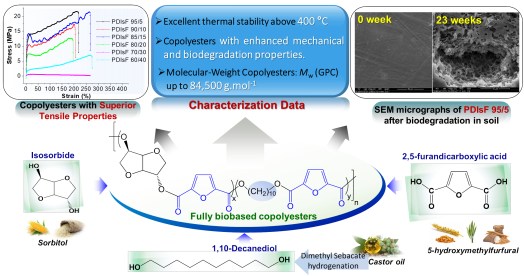
12 February 2019 Our recent study, published in ACS Sustainable Chemistry & Engineering “IF=8.198”, spotlighted a successful synthesis of a novel series of biobased poly(decamethylene-co-isosorbide 2,5-furandicarboxylate)s copolyesters from dimethylfuran-2,5-dicarboxylate, isosorbide, and 1,10-decanediol by melt polycondensation. These prepared materials revealed high sensistivity to the biodegradation process in soil, in addition to superior mechanical properties. Taking advantage of their features, PDIsFs have the potential to serve as promising and innovative biobased polymers for practical applications such as ecofriendly and sustainable plastic packaging.
10 February 2018 In this paper published in Polymer Degradation and Stability “IF=5.030”, we successfully synthesized by melt polycondensation a biobased Furanoate series of random poly(1,4-cyclohexanedimethanol-co-isosorbide 2,5-furandicarboxylate)s (PCIsFs) copolyesters. The prepared copolyesters showed good thermal stability. The efficiency of isosorbide insertion into polymer backbone was proved to be efficient route to enhance the thermal properties, for example significant increase in Tg, as well as to control the crystallinity and crystallization rate for PCIsFs copolyesters. It is noteworthy that, thanks to isosorbide, a correlation between PCIsFs chemical structure and their crystal structure and thermal characteristics were found…
For more details, click here